 Point to Remember: Mostly for transition and inner transition elements, the valence electrons are the electrons present in the shells outside the noble gas core. The periodic table and trends in valence electrons can be used to determine the number of bonds an atom is most likely to form. Atoms share electrons and form covalent bonds to satisfy the octet rule. (See the above or below periodic table to see the valence electrons of inner transition metals). 4.2: Covalent Bonds and the Periodic Table is shared under a CC BY-NC-SA 3.0 license and was authored, remixed, and/or curated by LibreTexts. Inner transition elements can have valence electrons ranging from 3 to 16. In some inner transition metals, the electrons of incomplete d-orbitals are also considered as valence electrons. Hence, for inner transition elements, the electrons of both f-subshells as well as s-subshell behave like valence electrons. The inner transition elements have incomplete f- subshells and they are very close to the outer s-subshell. (See the above or below periodic table to see the valence electrons of transition metals).Īlso the two bottom rows at the bottom of the periodic table are the inner transition elements (or f-block elements) also have the similar case. Hence, the transition elements (i.e d-block elements from group 3 to 12) can have more valence electrons ranging from 3 to 12. So, the electrons of both d-subshell and s-subshell behave like valence electrons. It is more difficult to find the valence electrons of transition elements as they have incompletely filled d-subshell and this d-subshell is very close to the outer s-subshell. What about valence electrons of transition and inner transition elements?įor the transition elements and inner transition elements, the case is more complicated. Well, this suits perfectly for the main group elements (i.e group 1, 2 and group 13 to 18), but what about the transition and inner transition elements? For example Example of group 1 All the elements of group 1 are highly reactive to. The elements lying in the same groups show similar chemical properties and they also have same number of valence electrons. Group 1 elements have just one valence electron and group 18 elements have eight, except for helium, which has only two electrons total. There are total 18 vertical columns on periodic table. In general, the number of valence electrons is the same within a column and increases from left to right within a row. Hence, magnesium has 2 valence electrons. Groups are the vertical columns on the periodic table. Here, you can see that the highest principal quantum number is 3, and the total electrons in this principal quantum number is 2. 
The electron configuration of magnesium is 1s 2 2s 2 2p 6 3s 2. Valence electrons can also be determined as the electrons present in the shell with highest principal quantum number (n). 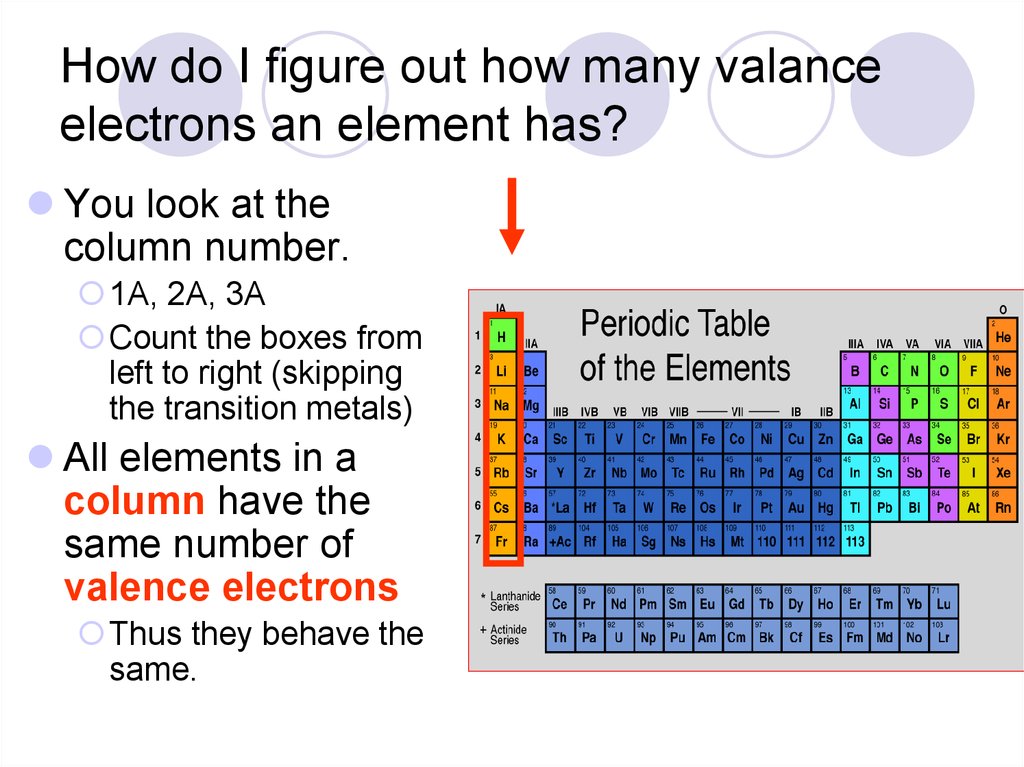
The magnesium element has 2 electrons in outermost orbit. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
